Project summary:
ASK1 (apoptosis signal-regulating kinase 1) is a MAP kinase kinase kinase (MAP3K) that controls various responses to oxidative and endoplasmic reticulum (ER) stress and calcium influx via the p38 and JNK signaling pathways. Deregulation of ASK1 activity is involved in the development of many diseases, including neurological disorders, amyotrophic lateral sclerosis, cardiovascular disease, diabetes and cancer. The catalytic activity of ASK1 is regulated through a complex mechanism that involves interaction with a variety of different binding partners including thioredoxin (TRX), TRAF factors and 14-3-3 proteins. Despite many years of intensive research, the molecular mechanism of ASK1 regulation remains unclear. The aim of this project is to elucidate the molecular basis of ASK1 regulation and to understand the role of ASK1 binding partners in its regulation. For this purpose, the structure of ASK1 complexes with different binding partners will be studied using integrative structural biology methods, in particular cryo-EM, SAXS, H/D exchange coupled to MS and chemical crosslinking coupled to MS. This is a project in the field of structural biology and biophysical chemistry of proteins and is funded by grants from the research group.
We offer:
- A 4-years Ph.D. student position starting from October 2023
- Funding from the Czech Science Foundation (GA ČR)
- Opportunity to gain expertise in preparation of recombinant proteins and methods of integrative structural biology
- Work in an international group of scientists
- Participation at international conferences and workshops
Requirements:
- M.Sc. or equivalent in Chemistry or Biochemistry (Beneficial: previous experience with molecular biology and/or preparation of recombinant proteins)
- Ability to communicate within an international team
- Interest in structural biology and protein biophysics
Five relevant publications of the research group:
- Mandal R., Kohoutova K., Petrvalska O., Horvath M., Srb P., Veverka V., Obsilova V., Obsil T. (2022) FOXO4 interacts with p53 TAD and CRD and inhibits its binding to DNA. Protein Sci. 31(5):e4287. doi: 10.1002/pro.4287
- Horvath M, Petrvalska O, Herman P, Obsilova V, Obsil T. (2021) 14-3-3 proteins inactivate DAPK2 by promoting its dimerization and protecting key regulatory phosphosites. Commun. Biol. 4(1):986. doi: 10.1038/s42003-021-02518-y
- Psenakova K., Hexnerova R., Srb P., Obsilova V., Veverka V., Obsil T. (2020) The redox-active site of thioredoxin is directly involved in apoptosis signal-regulating kinase 1 binding that is modulated by oxidative stress. FEBS J. 287(8):1626-1644. doi: 10.1111/febs.15101.
- Hagenbuchner J., Obsilova V., Kaserer T., Kaiser N., Rass B., Psenakova K., Docekal V., Alblova M., Kohoutova K., Schuster D., Aneichyk T., Vesely J., Obexer P., Obsil T., Ausserlechner M.J. (2019) Modulating FOXO3 transcriptional activity by small, DBD-binding molecules. eLife 8:e48876. doi: 10.7554/eLife.48876
- Alblova M., Smidova A., Docekal V., Vesely J., Herman P., Obsilova V., Obsil T. (2017) Molecular basis of the 14-3-3 protein-dependent activation of yeast neutral trehalase Nth1. Proc. Natl. Acad. Sci. U S A 114, E9811-E9820. doi: 10.1073/pnas.1714491114
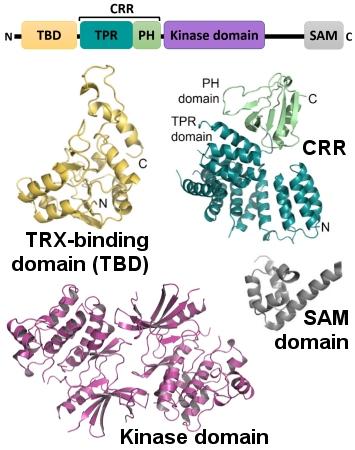
Domain structure of ASK1 with the structural models of individual domains.
Deadline is closed